 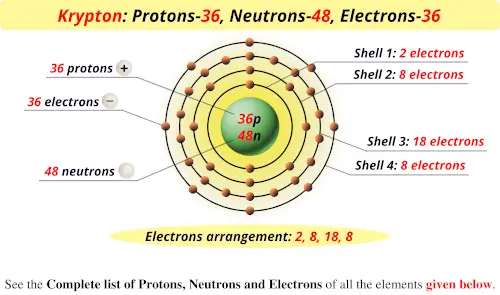 
So, the number of protons in the nucleus of an atom of Krypton is 36. Example: The element Krypton has the atomic number 36. The number of neutrons is determined by taking the atomic mass number (rounded to nearest whole number) and subtracting the atomic number from it. The atomic number provides this information for the atom. *Explain to students that all atoms have an overall neutral charge meaning they have the same number of protons and electrons. Instruct students that they will be constructing models of some of the elements using Playdough. Protons have a positive charge, electrons have a negative charge and neutrons have no charge therefore they are neutral. The particles are located inside and around the nucleus (center of each atom) of each atom. * After introducing students to the Periodic Table of Elements, explain that all these elements can be broken down to one single atom. Each atom is made up of these subatomic particles: protons, electrons and neutrons. Student copy of the Periodic Table of Elements Playdough (red, blue, green) Each student needs a small container of each color. This activity provides support for SOL 6.5a in which students are expected to create and interpret a simplified, modern model of the structure of an atom. This activity uses Playdough which is and always will be a favorite among kids. 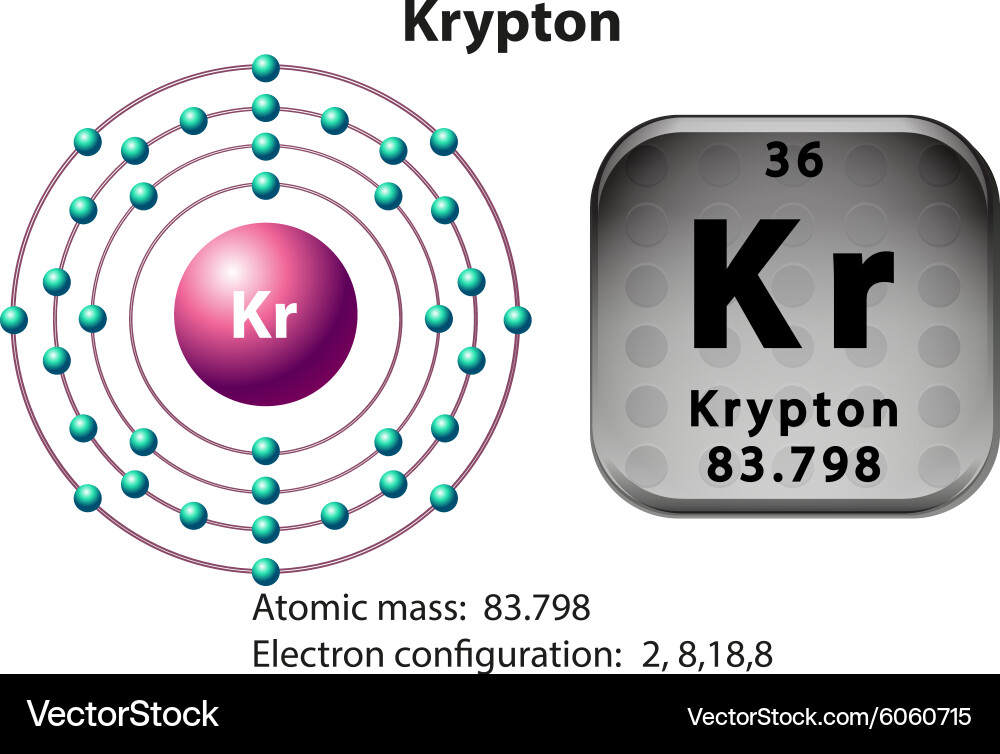
Overview: I created this lab activity to provide my students with a fun way to learn about the components of the atom. Students will investigate and understand that all matter is composed of atoms. Key ideas include atoms consisting of particles including electrons, protons and neutrons. This activity will be done after the introduction of the Periodic Table of Elements. 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
